 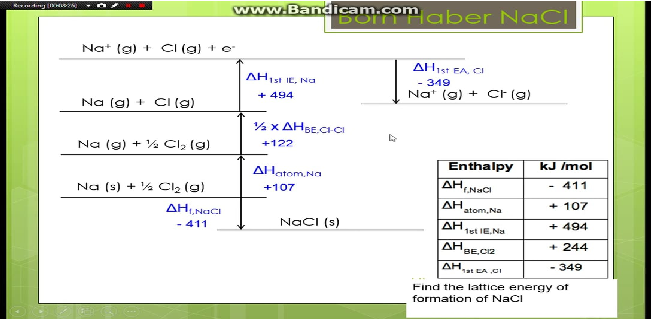
That means that we will have to use theoretical values of their lattice enthalpies. In 1918 Max Born and Alfred Landé proposed that the lattice energy could be derived from the electrostatic potential of the ionic lattice and a repulsive potential energy term. Let's look at this in terms of Born-Haber cycles of and contrast the enthalpy change of formation for the imaginary compounds MgCl and MgCl 3. It turns out that MgCl 2 is the formula of the compound which has the most negative enthalpy change of formation - in other words, it is the most stable one relative to the elements magnesium and chlorine. U, which is always a positive number, represents the amount of energy required to dissociate 1 mol of an ionic solid into the gaseous ions. Lattice energy is the amount of energy released when. The lattice energy of nearly any ionic solid can be calculated rather accurately using a modified form of Equation 4.2.1: (4.2.1) U k Q 1 Q 2 r 0, w h e r e U > 0. LatticeH denotes the molar lattice enthalpy. Where: LatticeU denotes the molar lattice energy. The question arises as to why, from an energetics point of view, magnesium chloride is MgCl 2 rather than MgCl or MgCl 3 (or any other formula you might like to choose). This chemistry video tutorial provides a basic introduction into the lattice energy of ionic compounds. The following equation may be used to represent the molar lattice energy of an ionic crystal in terms of molar lattice enthalpy, pressure, and volume change: LatticeU LatticeH pVm. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
